With an estimated 100 million types and so abundant that all the life forms put together, the viruses were discovered only after the invention of the electron microscope in 1939. The subsequent research o their structure, diversity and characteristics help humanity to understand the havoc they had wrecked on mankind throughout history. While some of the diseases that virus have been causing have been taken care of but the challenge remains as these RNA strands are evolving with every passing day. HIV, Ebola, Dengue fever, SARS and MEARS are the recent epidemics the virus has created in anticipation of the Coronavirus trigged Covid-19 pandemic. In these is passages excerpted from Viruses: A Very Short Introduction, Dorothy H Crawford, a professor of medical biology at the University of Edinburgh explains the recent epidemics preceding the one that has brought the world on its knees:
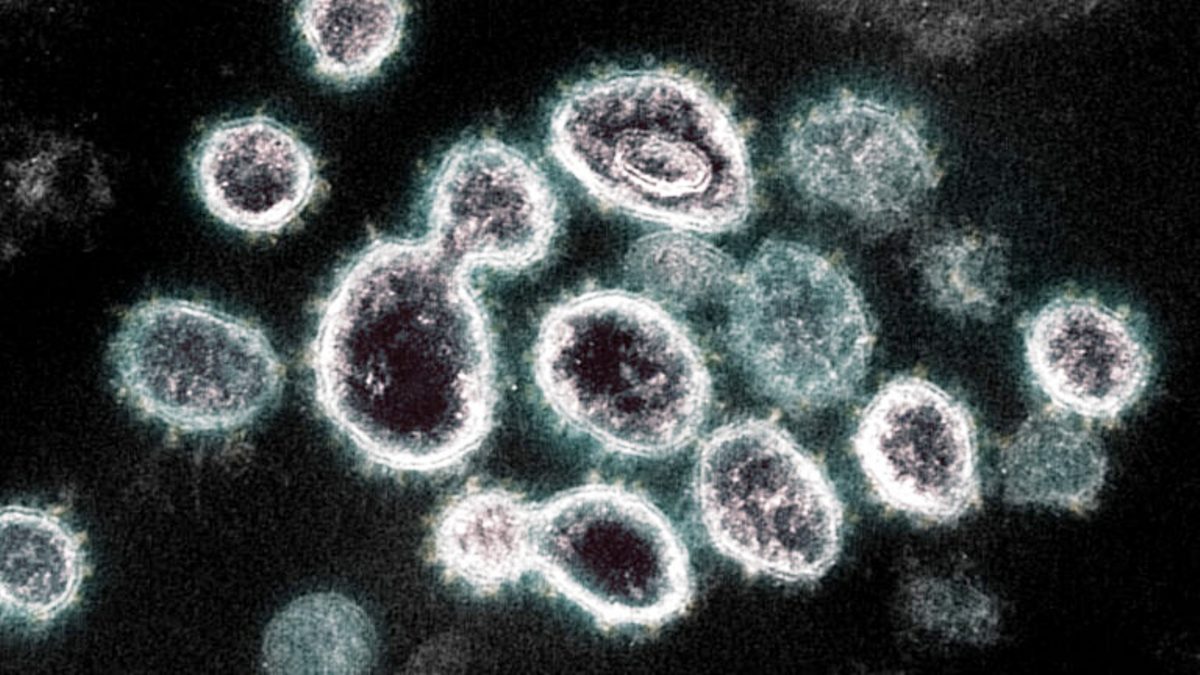
SARS (Severe acute respiratory syndrome) coronavirus first emerged in November 2002 in Foshan, Guangdong Province, China, where it caused an outbreak of atypical pneumonia. Initially, the virus spread locally, particularly among patients’ family members and hospital staff, but everything changed in February 2003 when a doctor who had treated SARS cases in Guangdong Province unwittingly carried the virus to Hong Kong. He stayed one night at the Metropole Hotel in Hong Kong before being admitted to hospital, where he died of SARS a few days later. In the hospital, the virus spread to staff, which sparked the Hong Kong epidemic.

During his 24-hour stay in the hotel, the doctor transmitted the virus to at least 17 guests (apparently he sneezed in the lift), who then carried it to 5 more countries, thus spawning epidemics in Canada, Vietnam, and Singapore. This rapid dissemination of the virus threatened to cause a pandemic, but surprisingly by July 2003, it was over, the final toll being around 8000 cases and 800 deaths involving 29 countries across 5 continents.
SARS coronavirus spreads through the air and causes disease in almost everyone it infects. After an incubation period of 2 to 14 days, victims develop fever, malaise, muscle aches, and a cough, sometimes progressing rapidly to viral pneumonia that requires intensive care, with mechanical ventilation in around 20% of cases. But with no known treatment or preventive vaccine, how was the epidemic conquered so effectively?
Left to its own devices, SARS coronavirus would undoubtedly have continued its trail of destruction but, fortunately, many of its characteristics played into the hands of those trying to stop it, and contributed to its speedy demise. Importantly, the virus mostly causes overt disease, with few unidentified silent infections. This meant that cases and their contacts could be recognized and isolated, and since victims are only infectious once the symptoms have developed, this prevented further spread. Also, as the disease is usually severe and debilitating, relatively few patients, excepting the doctor from Guangdong, travelled far while infectious.
During SARS, the virus is produced in the lungs and spread by coughing. This generates relatively heavy mucus droplets that do not spread far through the air; hence close contacts like family members and hospital staff are mainly at risk, the latter constituting over 20% of cases worldwide. Once all these factors were appreciated, old-fashioned barrier nursing and isolation of patients and their contacts were enough to interrupt virus spread and prevent a pandemic.
SARS, HIV, H5NI
Unlike SARS coronavirus, HIV has been spreading among humans since the early 1900s and despite drugs, which control the infection; it is still on the increase in certain areas of the world. Currently, there are 33 million people living with it. HIV has caused over 25 million deaths since the first report of AIDS in 1981. It is interesting to examine the reasons for this lack of control, and to contrast these with the success of the SARS control programme.

Firstly, although SARS coronavirus had spread internationally by the time it was recognized by the World Health Organization (WHO), it had only infected humans for a few months. Compare this to the estimated 100 years during which HIV was silently creeping around sub-Saharan Africa, where poverty, wars, and poor health services conspired to facilitate its spread, and prevent the recognition of AIDS as a new disease.
Secondly, in contrast to SARS’ short incubation period and infectivity coinciding with overt disease, HIV has an average asymptomatic period of eight to ten years, and during this time the carrier may transmit the virus to any number of contacts.
Thirdly, the two viruses spread by completely different means. Whereas SARS coronavirus’s airborne flight can easily be intercepted, interruption of HIV’s transmission is more problematic. HIV spreads most commonly by sexual contact. Other routes of spread include mother to child during birth and breast-feeding, in transplanted organs, transfused blood and blood products, and via contamination of surgical instruments as well as injecting drug users’ equipment. These non-sexual routes can, in theory, be interrupted, but they are almost insignificant in global terms compared to its spread via heterosexual contact. In exploiting the basic human urge to procreate, HIV targets the young and sexually active and is passed unwittingly from one apparently healthy host to another through sexual networks. Although its transmission can be halted by barrier devices, the vast amounts of money spent on the promotion of condom use for safer sex have not altered sexual practices sufficiently to halt the pandemic.
On occasions, viruses do enter and successfully replicate in cells of a new host species, but after a window of opportunity lasting about a week during which they can colonize the host and reproduce, their offspring must move on to another susceptible host before the developing host immunity wipes them out. SARS coronavirus and H5N1 (bird) flu have both managed to infect humans but differ in their success to date. Whereas SARS coronavirus can spread between humans, H5N1 flu, which first jumped from birds to humans in 1997, is unable to do so. This flu virus strain is still poorly adapted to its new (human) host, and we will be in danger of an H5N1 flu pandemic only once it evolves an efficient method of spreading between us.
Flu Viruses
Flu is a prime example of a virus that mutates frequently, a process called antigenic drift. The flu virus circulates constantly in the community, accumulating genetic changes and causing regular winter outbreaks and larger epidemics every eight to ten years. However, its story is actually much more complicated. There are three flu strains, A, B, and C, and flu A is a zoonotic virus. With the help of wild birds, this virus can also undergo recombination, or antigenic shift, producing an entirely new strain of flu in one go by exchanging fragments of its genome with other strains. This has the potential to cause a pandemic.
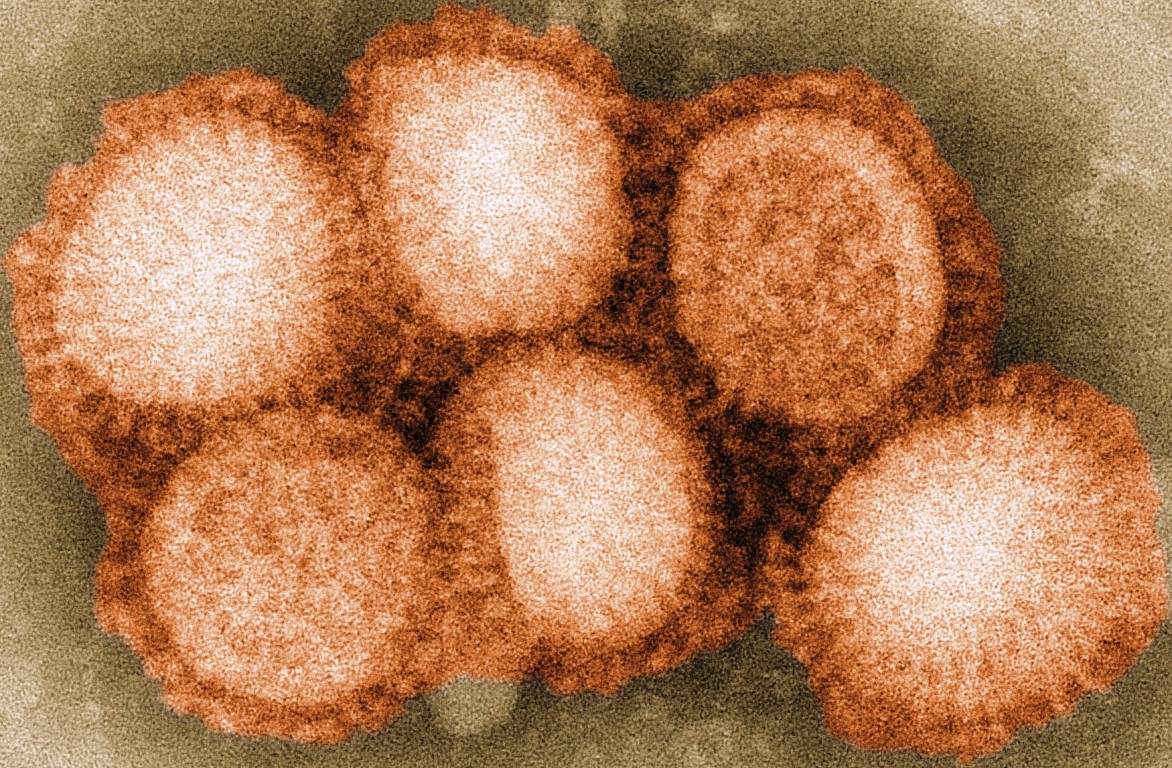
The natural hosts of flu A viruses are aquatic birds, particularly ducks, but the viruses also infect a variety of other animals including domestic poultry, pigs, horses, cats, and seals. Flu A replicates in birds’ guts and is excreted in their faeces, causing no symptoms but effectively spreading to other bird populations. Flu viruses have eight genes, which are segmented, meaning that instead of its genome being a continuous strand of RNA, each gene forms a separate strand. The H (haemagglutinin) and N (neuraminidase) genes are the most important in stimulating protective host immunity. There are 16 different H and 9 different N genes, all of which can be found in all combinations in bird flu viruses. Because these genes are separate RNA strands in the virus, on occasions they become mixed up, or recombined. So if two flu A viruses with different H and/or N genes infect a single cell, the offspring will “carry varying combinations of genes from the two-parent viruses. Most of these viruses will not be able to infect humans, but occasionally a new virus strain is produced that can jump directly to humans and cause a pandemic, as we have experienced recently with swine flu.
Over the last century, there have been five flu pandemics: in the H1N1 ‘Spanish’ flu of 1918, all eight genes came from birds; the H2N2 ‘Asian’ flu of 1957 acquired three new genes, including H and N from birds; and the H3N2 ‘Hong Kong’ flu of 1968 acquired two new genes from wild ducks. The ‘Russian’ flu of 1977, which probably escaped from a lab in Russia, was a 1950s version of H1N1; whereas the H1N1 ‘swine’ flu, which appeared in Mexico in 2009, has six genes from North American and two genes from Eurasian pig flu viruses.
Species Jump
The transfer of ‘new’ zoonotic viruses from their primary host to humans can be facilitated by certain behaviours or cultural practices, and we now know that a particular risk is our interaction with wild animals, many of which carry viruses with the potential to infect us. Both HIV and SARS coronavirus were introduced into the human population when their natural hosts were hunted and killed for consumption.
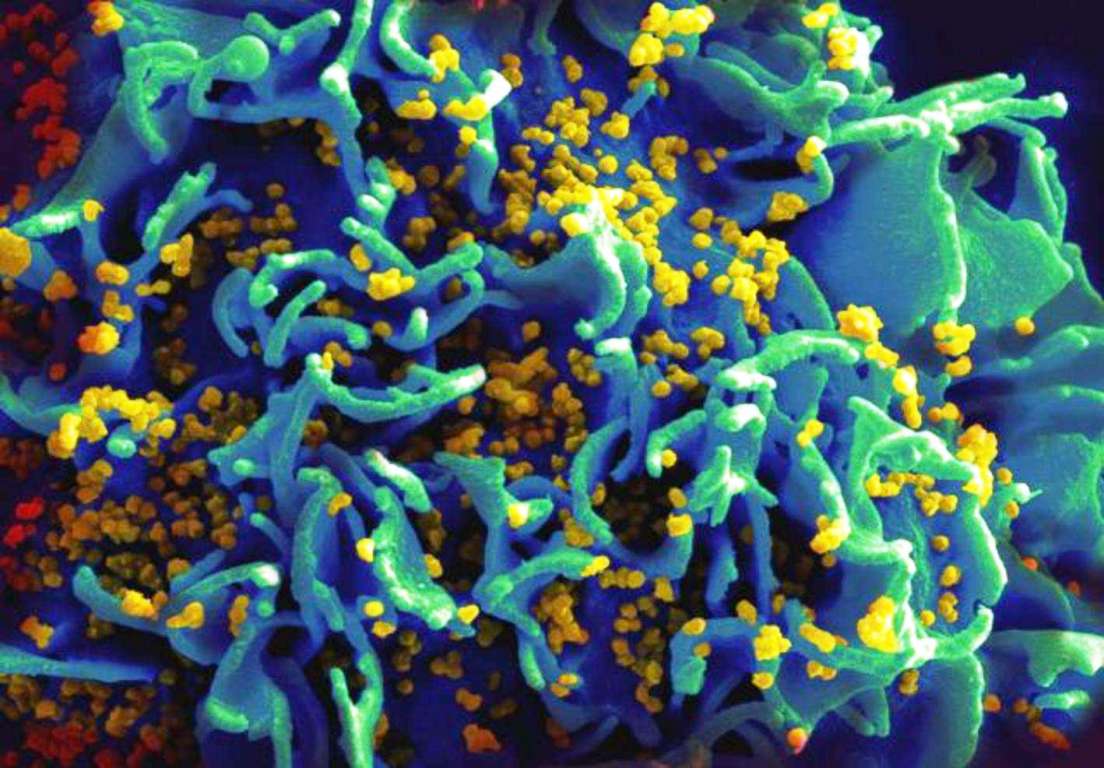
It is now clear that HIV-like viruses have jumped from primates to humans in central Africa on several occasions and that one of these viruses, HIV-1 type M, has succeeded in spreading globally. The ancestor of this virus has been traced to a subspecies of chimpanzees (Pan troglodytes troglodytes), among whom it can cause an AIDS-like disease. Since these animals are hunted for bushmeat, it is most likely that human infection occurred by blood contamination during the killing and butchering process. This transfer took place some 100 years ago, probably in southeast Cameroon where the chimpanzees carrying the virus most similar to HIV-1 type M live. Scientists postulate that the virus (inside humans) travelled from Cameroon along the Sangha River, a tributary of the Congo River, to reach Leopoldville (now called Kinshasa), then the capital of the former Belgian Congo, from where it spread globally.
SARS coronavirus also entered the human population from an animal food source, this time in the live animal markets of China. Here, there are a number of small mammals on offer and several, most noticeably the Himalayan palm civet cat, carry SARS-like viruses. As the natural reservoir of SARS coronavirus has now been identified as the fruit bat, it is presumed that the virus transferred to other animal species in markets where they are packed into overcrowded cages, and then jumped to the market traders.
Ebola
SARS is not the only potentially lethal virus carried by bats; several bat species are reservoirs for viruses that have recently jumped to humans. In fact, bats almost certainly transmit the much-feared and highly infectious Ebola and Ebola-like viruses. Epidemics of Ebola viral haemorrhagic fever hit rural populations in central Africa from time to time, and these outbreaks have increased in frequency in DRC, Gabon, and Sudan since the mid-1990s. Ebola virus was discovered after an explosive outbreak in Yambuku, a remote village in northern Zaire (now DRC), in 1976, and was named after the local Ebola River. This epidemic began with a schoolteacher who developed a headache and fever after returning from a trip into the bush. He was treated for malaria at the local mission hospital, but his symptoms progressed to a full-blown viral haemorrhagic fever with soaring temperature, severe abdominal pain, diarrhoea, vomiting, muscle cramps, and generalized bleeding. He died within a few days. The virus, transmitted by direct contact with the patient and his body fluids, then spread to his family, other hospital patients, and staff, eventually infecting 318 people in the village and killing 280 of them.”
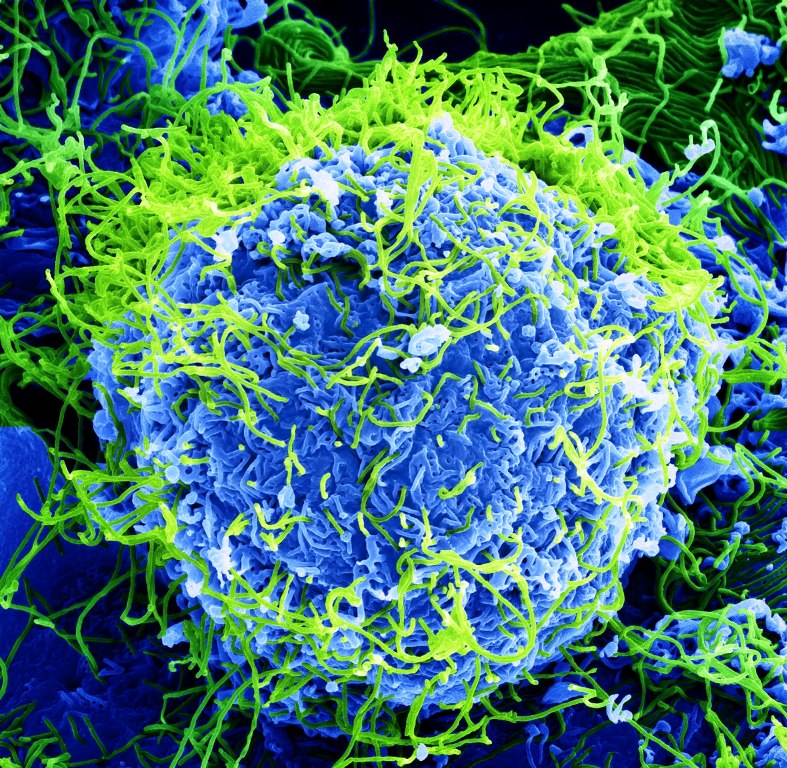
Unfortunately, the virus has recently jumped to large apes, particularly chimpanzees and lowland gorillas. This not only threatens the very existence of these endangered species but also provides an additional transmission route to humans when they come into contact with these animals, perhaps accounting for the recent reported rise in outbreaks.
Nipah Virus
Another dangerous bat-transmitted virus emerged in 1997 when a group of Malaysian farmers reported a respiratory disease outbreak among their pigs, and later several pig farmers and abattoir workers came down with encephalitis. Fortunately, the disease did not spread directly from person to person and was later controlled by slaughtering over a million pigs in 1999. Sadly, by this time, there had been 265 cases of encephalitis with 105 fatalities. A novel paramyxovirus was isolated from a victim’s brain and named Nipah virus after the village in which he lived. The virus was traced to “fruit bats, and its trail to humans probably began when a colony of bats was left homeless by deforestation. The bats relocated to trees near the pig farms and the virus spread to the pigs via bat droppings, and then from the pigs to the farmers and abattoir workers.
Due to our invasion of their territories, bats and humans are coming into contact with increasing frequency. The Nipah virus turns out to be very similar to bat-borne Hendra virus, isolated in 1994 from the victims of an outbreak of severe respiratory disease on Hendra farm in Brisbane, Australia, where it killed 14 horses and one of their trainers. Similar outbreaks in West Bengal in 2001 and in Bangladesh in 2001 and 2004 are also attributed to bat viruses, indicating that these cute, furry animals are far from safe companions.
(The book was published by Oxford University Press in 2011 and is globally being considered a crash course in understanding the world of viruses.)














