An Indian biotech start-up has secured a global first: a patented, third-generation CAR-T therapy that targets cancer with unprecedented precision—offering new hope for affordable, life-saving treatment, reports Humaira Nabi

In a significant leap for Indian biotechnology, Delhi-based start-up Cellogen Therapeutics has secured the patent for the world’s first indigenously developed bispecific, third-generation Chimeric Antigen Receptor T-cell (CAR-T) therapy. The development represents a momentous advance in cellular immunotherapy, one that holds the potential to revolutionise treatment for blood cancers such as leukaemia, lymphoma, and multiple myeloma.
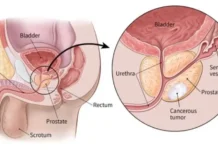
CAR-T therapy, a form of personalised immunotherapy, involves extracting a patient’s T cells—white blood cells critical to immune defence—and genetically modifying them to express synthetic receptors capable of targeting specific antigens on cancer cells. Once reintroduced into the patient’s body, these reprogrammed T cells (CAR-T cells) actively seek and destroy malignant cells.
Although CAR-T therapies have been in use globally for over a decade, challenges remain: T-cell exhaustion, limited persistence in the body, and relapse due to antigen escape have all hindered long-term efficacy. Cellogen’s third-generation innovation aims to overcome these limitations by introducing bi-specific targeting and enhanced T-cell durability.
The Science of Precision
Cellogen’s third-generation CAR-T platform integrates multiple co-stimulatory domains—CD28, 4-1BB, and OX40—enhancing the activation, proliferation, and survival of T-cells. Crucially, the therapy also includes two distinct single-chain variable fragments (scFvs) for antigen recognition. This bi-specificity allows CAR-T cells to target two tumour-associated antigens simultaneously, a feature that directly addresses one of the most serious shortcomings of earlier CAR-T therapies: antigen escape.
In previous treatments, cancer cells often mutated or down-regulated targeted antigens, thereby evading detection and causing disease relapse. By simultaneously targeting CD19 and CD20 antigens on B cells, Cellogen’s innovation enhances the breadth and durability of immune response against B-cell malignancies.
This platform is the result of years of rigorous research and development, said Dr Tanveer Ahmad, a Kashmir-born researcher and Assistant Professor at Jamia Millia Islamia University, Delhi, who led the project. He noted that the therapy, developed with state-of-the-art cell engineering techniques, represents a significant step forward in improving patient outcomes.
Dr Areej Akhtar, a PhD graduate from Jamia Millia Islamia who led the study under Dr Ahmad’s supervision, emphasised that their product stands apart from earlier CAR-T platforms currently available in the United States and under development in India. This is the first third-generation construct to enter clinical trials in the country, she explained.

Minimising Risk, Maximising Response
Alongside its enhanced tumour-killing capacity, Cellogen’s therapy mitigates life-threatening side effects commonly associated with CAR-T, such as cytokine release syndrome (CRS) and neurotoxicity. These complications, often resulting from uncontrolled immune activation, have posed serious risks in previous treatments. By refining immune signalling, the new platform enables more regulated immune responses, potentially reducing adverse events and improving safety profiles.
Cellogen’s innovation is not an isolated achievement but builds upon a broader body of rigorous scientific inquiry. A recent preclinical study published in Cell Reports Medicine introduced an experimental fourth-generation CAR-T construct developed by the same team. This construct incorporates GLP-1 receptor agonists and Urolithin A—a naturally occurring compound known to enhance cellular metabolism. Apart from Dr Tanvir (guide) and Areej, other scholars who worked on the project included Mohammad Shakir,
Mohammad Sufyan Ansari, Soumya Sinha Roy, and Irfan Rahman.
“We screened twenty-seven drugs across 3,357 combinations and discovered that GLP-1 and Urolithin A synergistically up-regulated autophagy and mitophagy,” Dr Akhtar said. These cellular processes—essential for removing damaged components—also prevent T-cell exhaustion, thereby increasing therapy longevity. In murine models, the combination demonstrated a marked increase in CAR-T cell persistence and anti-tumour activity.
While fourth-generation therapy remains in preclinical stages, the bispecific third-generation construct has now progressed to clinical trial readiness. The early results are highly promising, Dr Akhtar added, but human trials take time. “We are closely monitoring whether the persistence seen in animal models translates to similar outcomes in humans.”
Making Cancer Care Affordable
One of the principal barriers to CAR-T therapy in India has been its prohibitive cost. Globally, such treatments often exceed Rs 3–5 crore per dose, rendering them inaccessible to the majority of Indian patients.
Cellogen aims to break that barrier. “Our goal is to reduce the cost to rs 15–20 lakh per dose,” said Dr Akhtar. The company believes that if local manufacturers can produce reagents currently imported from abroad, costs could fall even further.
To support its clinical translation, Cellogen has secured two million US dollars in funding from NATCO Pharma Limited, which has taken a 5.38 per cent equity stake in the company. The therapy is now set to undergo trials in partnership with Christian Medical College (CMC), Vellore—one of India’s foremost medical institutions.
Expanding Horizons
Although the present focus remains on B-cell cancers, the underlying technology may also prove effective against solid tumours, which have long presented formidable challenges for CAR-T therapies. The complexity of the tumour microenvironment and the difficulty of T-cell infiltration have limited progress in this area. However, Cellogen’s innovations in persistence and bi-specific targeting offer new pathways for therapeutic expansion.
The company is currently collaborating with CMC Vellore and other leading institutions to expedite clinical trials, ensuring that the therapy is rigorously tested for both safety and efficacy. If successful, it could pave the way for broader applications in oncology, particularly in regions where high-cost therapies have remained out of reach.
Cellogen Therapeutics’ breakthrough situates the company at the forefront of next-generation cancer treatment. With its emphasis on scientific precision, affordability, and therapeutic durability, this innovation could redefine cancer care—not only in India but across the globe. As clinical trials unfold, the promise of a more accessible, life-altering therapy inches closer to reality.
A Happening Laboratory
Jamia’s laboratory – Multidisciplinary Centre for Advanced Research and Studies (MCARS)- has remained consistently in the news for innovations and newer things. In 2024 fall, it achieved a breakthrough in oral cancer detection with the development of an AI-driven diagnostic technology. Led by Tanveer Ahmad and his PhD scholar Ms Nisha Chaudhary, the innovation integrates AI and digital pathology to enhance accuracy, affordability, and accessibility in cancer diagnostics.
Recognised by the Government of India, the system was awarded a patent on December 24, 2024, for its ability to analyse tissue images and genomic markers to predict and classify oral malignant disorders, including OSMF, OLL, OLP, and OSCC. It also assesses the severity of OSCC and predicts precancerous conditions’ progression, enabling early intervention.
Published in Scientific Data (Nature Publishing Group), the research provides a high-resolution AI image dataset valuable to global scientists. The project, involving eminent researchers, has positioned JMI at the forefront of digital pathology and cancer research.
Two Kashmiri Scientists In News
Interestingly, there are two Kashmiri scientists who have been in the news for cancer treatment, almost using the same technology. One is Jamia faculty, Dr Tanveer, a gene-editing expert who led multiple innovations, including a COVID-19 detection tool, and advancing life-threatening disease diagnostics. Another is Dr Khalid Shah, currently the Vice Chairman of Research at Harvard Medical School and Director of the Centre for Stem Cell and Translational Immunotherapy.
In lathe st few years, Dr Shah had two major breakthroughs. In 2021, his groundbreaking work in cancer cell therapy was recognised at the World Medical Innovation Forum. His innovative approach, which encourages cancer cells to turn against tumours, was featured among the Disruptive Dozen – 12 emerging gene and cell therapy (GCT) technologies with the greatest potential to revolutionise healthcare. His research focused on Glioblastoma, an aggressive brain cancer, and leverages CRISPR technology to genetically reprogram tumour cells for targeted treatment. Early this year, he did a follow-up breakthrough that was published in the Journal of Clinical Investigation. It was about his development of a safety mechanism to eliminate cancer-killing cells after they have served their purpose. The new “safety switch” uses the RIPK3 enzyme to trigger programmed cell death, enhancing the safety and effectiveness of cell-based therapies.
Laboratory models showed improved tumour suppression and immune response, offering new hope for glioblastoma treatment. Dr Shah holds over fifteen patents and has founded two biotech companies to translate his discoveries into clinical applications.